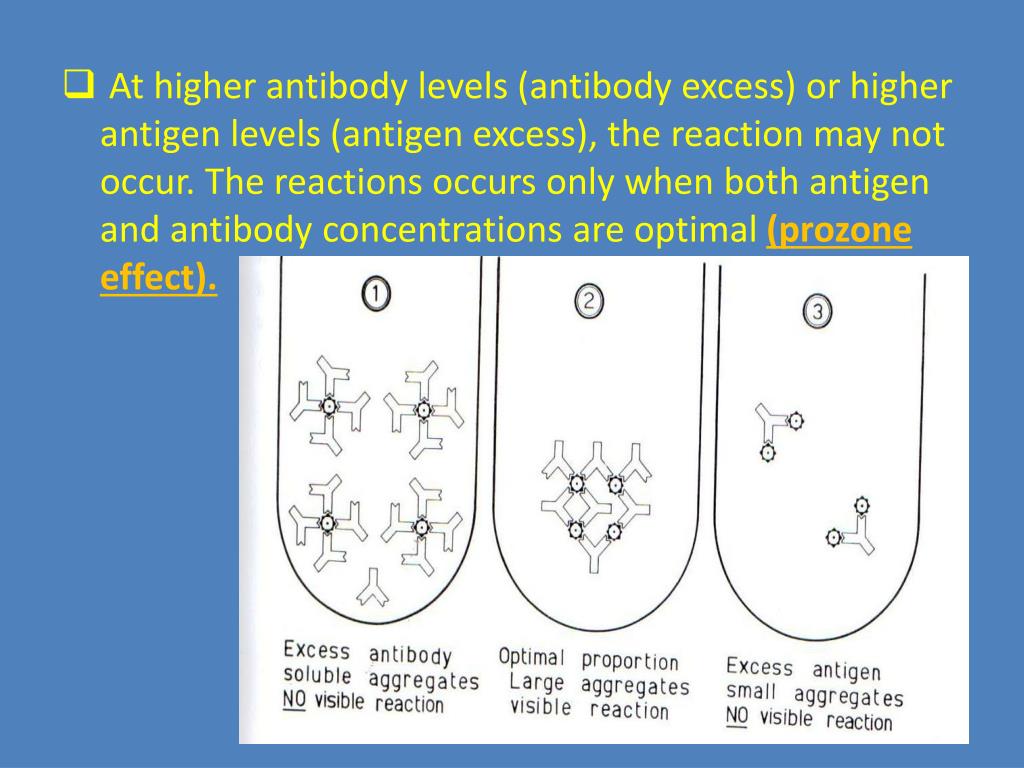
Serologic diagnostics of SARS-CoV-2 and co-existing pathogens in Benin. We used previously described recombinant S-based immunofluorescence assays ( 7) to test for specific antibodies to common cold betacoronavirus human coronavirus (HCoV) OC43 and HCoV-HKU1.įigure 1. Plaque-reduction neutralization tests (PRNTs) were performed by using similar methods for SARS-CoV-2 and ZIKV as described ( 4, 6). Serum samples also were tested by using commercially available ELISA kits ( Euroimmun) against the Zika virus (ZIKV) nonstructural protein 1 (NS1) antigen (IgG), the Epstein-Barr virus (EBV) nuclear antigen 1 (EBNA1) (IgG), and the EBV viral capsid (CA) antigen (IgM and IgG), as well as real-time PCR tests (TIB MOLBIOL, ) for all human pathogenic Plasmodium species, EBV, and cytomegalovirus (CMV). We also used the SCoV-2 Detect IgG ELISA (InBios, ), an IgG-only S1 antigen-based test authorized for emergency use by the US Food and Drug Administration. We tested all 68 serum samples by using commercially available ELISAs from EUROIMMUN ( ) that rely on different antigens and antibody classes: SARS-CoV-2 nucleocapsid (N) antigen (IgG), spike 1 (S1) subunit (IgG and IgA), and Middle East respiratory syndrome coronavirus (MERS-CoV) S1 (IgG). Sampling was approved by the ethics committee of the Benin Ministry of Health (approval no. We also included 60 serum samples from patients with acute febrile illness tested as part of hemorrhagic fever surveillance during October–November 2019 as prepandemic controls ( Table 2). The average sampling time was 8 (range 1–10) days after RT-PCR confirmation of SARS-CoV-2 infection ( Table 1). We obtained convalescent serum samples from 8 patients in Benin with RT-PCR–confirmed COVID-19 during March–April 2020. We conducted a SARS-CoV-2 serologic assessment in Benin by using samples from patients with RT-PCR–confirmed SARS-CoV-2 infection and controls sampled before the first SARS-CoV-2 detection in March 2020. 
However, many serologic tests have not been validated in resource-limited settings ( 5). Serologic studies in Europe and Asia indicate high sensitivity and specificity of widely used SARS-CoV-2 antibody ELISAs ( 3, 4). In addition, antibody-based serologic testing is a valuable epidemiologic tool to assess COVID-19 spread and potential immunity to SARS-CoV-2. In later stages of disease, antibody-based serologic testing can complement diagnosis of SARS-CoV-2 infection. Diagnosis of the causative pathogen, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is based on reverse transcription-PCR (RT-PCR) to detect viral nucleic acid or serologic assays to detect SARS-CoV-2 antigens in early stages of disease ( 1, 2). Since its emergence in China late 2019, coronavirus disease (COVID-19) had caused >41 million cases and >1.1 million deaths globally by October 2020, according to the World Health Organization ( ). Nagel) German Centre for Infection Research, associated partner Charité-Universitätsmedizin, Berlin, Germany (J.F. Djingarey) Deutsche Gesellschaft für Internationale Zusammenarbeit, Bonn, Germany (M. Joël Aïssi) World Health Organization Regional Office for Africa, Health Emergencies Programme, Brazzaville, Congo (M.H. Hounkpatin) Conseil National de Lutte contre le VIH-Sida, la Tuberculose, le Paludisme, les IST et les Epidémies, Cotonou (M.A. Badou) Charité-Universitätsmedizin Berlin, corporate member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, Berlin, Germany (A.-L. Yadouleton) Laboratoire des Fièvres Hémorragiques Virales du Benin, Cotonou (A. Author affiliations: Université Nationale des Sciences, Technologies, Ingénierie et Mathématiques, Cotonou, Benin (A.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
